Recently, a research team led by Prof. LIN Yao-Xin and Prof. WANG Hao, in collaboration with Prof. GAO Yurui from the National Center for Nanoscience and Technology (NCNST) of the Chinese Academy of Sciences, published a study in Nature Biomedical Engineering. The team successfully developed a novel artificial intelligence (AI) model to optimize ionizable lipids, addressing two long-standing bottlenecks in mRNA drug delivery: low efficiency and a lack of precise organ targeting.
Traditionally, LNP design has primarily focused on the two-dimensional (2D) chemical structures of ionizable lipids, often overlooking their dynamic three-dimensional (3D) spatial conformations in physiological environments. This oversight has limited the in-depth understanding and precise design of LNP. In this study, the research team introduce a new approach that incorporates 3D spatial conformation as a key predictive factor into an AI model.
The research team first built a library of ionizable lipids and used molecular dynamics (MD) simulations to capture the dynamic conformation of each lipid. After converting these 3D conformational data into 2D density images to train the AI model, the AI-guided screening successfully identified lipid P1. Compared to the approved lipid ALC-0315, lipid P1 exhibited a 14.8-fold increase in mRNA delivery efficiency. Notably, the unique 3D structure of lipid P1 allows it to specifically adsorb immunoglobulin M (IgM) in the blood, thereby enabling precise spleen-targeted delivery.
Utilizing this spleen-targeting LNP, the team developed an mRNA cancer vaccine that exhibited significant therapeutic efficacy in a mouse melanoma model. The vaccine successfully activated potent T-cell immune responses and induced high levels of antigen-specific antibodies, achieving dual activation of both humoral and cellular immunity. Ultimately, the vaccine effectively regressed established tumors and provided a long-term immune protection in mice.
This study pioneers a new design logic for ionizable lipids, shifting from the empirical exploration of 2D chemical structures to the AI-based precise analysis of 3D spatial conformations. Furthermore, the research elucidates the molecular mechanism by which ionizable lipids achieve lysosomal escape during mRNA delivery. By revealing potential methods to overcome the liver enrichment of mRNA nanomedicines, this work provides an efficient and safe delivery solution for cutting-edge fields such as gene editing, protein replacement therapy, and in vivo CAR-T therapy.
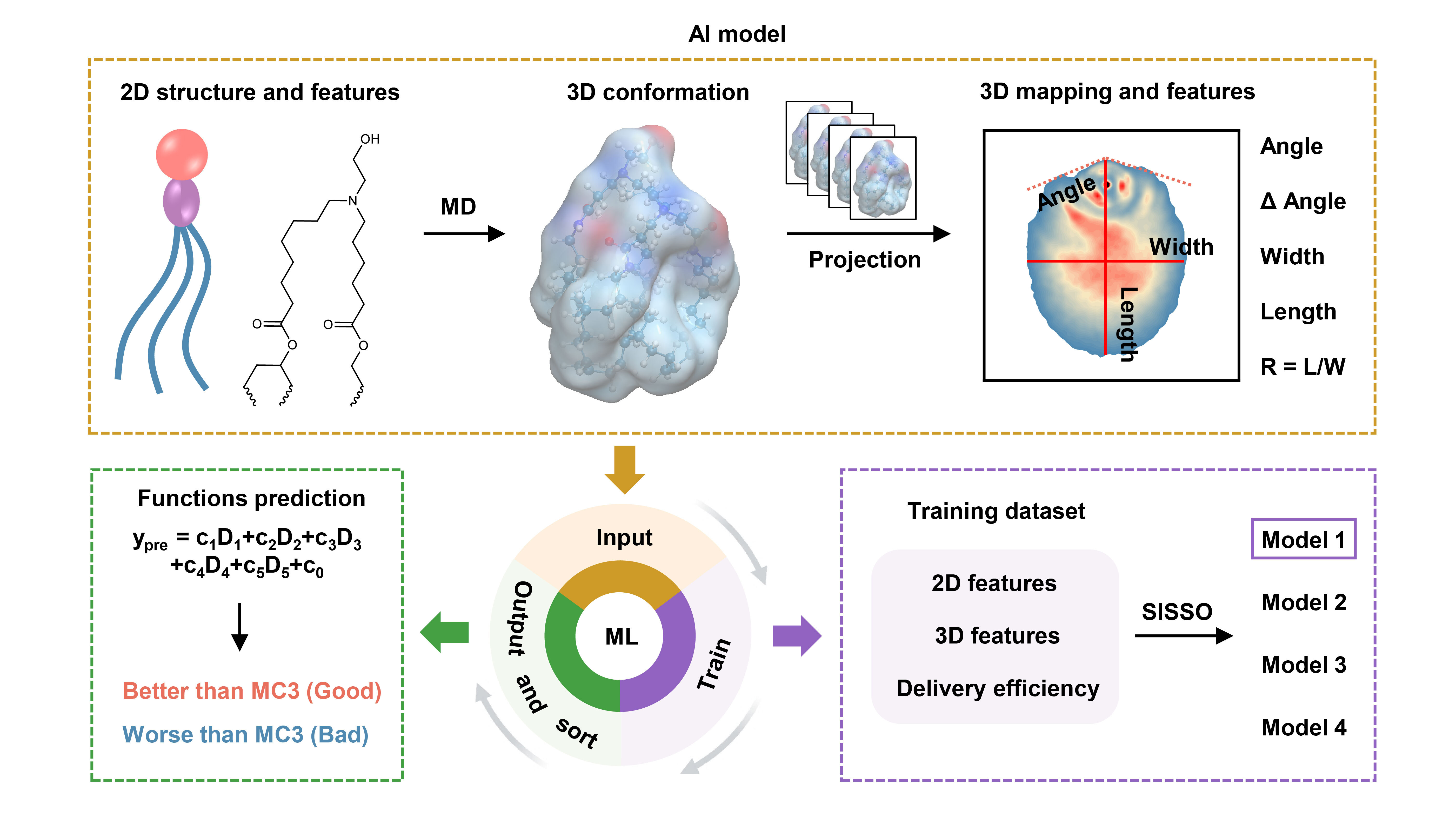
Schemetic illustration for the development of an AI model for LNP optimization. (Image by SU Linjia et al.)
Contact:
Prof. Lin Yao-Xin
National Center for Nanoscience and Technology (NCNST)
E-mail: linyx@nanoctr.cn




