In a studypublished in Nature Synthesis, a research team led by Prof. ZHAO Shenlong from the National Center for Nanoscience and Technology (NCNST) of the Chinese Academy of Sciences has developed ultrafine high-entropy alloy nanowires (UF-HEANWs) as highly efficient and durable electrocatalysts for the direct electrosynthesis of chlorine from seawater, offering a low-cost and energy-efficient alternative to the century-old chlor-alkali process.
Chlorine (Cl2) is an indispensable industrial chemical, underpinning the production of pharmaceuticals, disinfectants, and polymers such as polyvinyl chloride. However, more than 95% of global chlorine production currently relies on the conventional chlor-alkali process, which consumes large amounts of electricity, requires highly purified brine, and contributes substantially to carbon emissions.
To address these drawbacks, the researchers designed one-dimensional (1D) UF-HEANWs composed of Pt, Ni, Co, Fe, and Mo. Owing to their ultrathin morphology and lattice-distorted structure, the nanowires are enriched with abundant atomic steps and unsaturated coordination sites, which are highly favorable for the chlorine evolution reaction (CER). When integrated into a seawater flow electrolyzer, the UF-HEANW anodes achieved a chlorine selectivity of 98.1% at an industrial-scale current density of 10 kA m-2, while maintaining continuous operation for over 5,500 hours. Even under harsh acidic seawater conditions at 80 °C, the system exhibited negligible performance degradation and minimal platinum dissolution, demonstrating exceptional long-term durability.
Operando spectroscopic studies revealed that the outstanding performance originates from the dynamic formation of high-valent Pt–O active sites during operation. The lattice distortion and atomic-step features of the high-entropy nanowires induce localized electronic heterogeneity, which promotes chloride adsorption, facilitates chlorine desorption, and effectively suppresses competing oxygen evolution reaction (OER).
Beyond electrochemical performance, a comprehensive techno-economic analysis highlighted the practical advantages of the new system. Direct chlorine electrosynthesis from seawater using high-entropy alloy (HEA) anodes reduced the total production cost to US$463.7 per ton of chlorine which is 32.8% lower than that of the conventional chlor-alkali process. The cost reduction is primarily attributed to a 51.3% decrease in electrolysis energy consumption via HEA anodes and an 83.1% reduction in feedstock costs by replacing purified brine with abundant seawater. "This work demonstrates a viable pathway for sustainable chlorine production by coupling advanced HEA nanomaterials with seawater electrolysis," said Dr. YANG Yongchao, the first author of this work.
This finding not only provides a scalable and economically competitive alternative to traditional chlor-alkali technologies, but also highlights the transformative potential of high-entropy materials in addressing key challenges at the energy-water nexus, particularly in harnessing Earth-abundant seawater resources for carbon-neutral chemical synthesis.
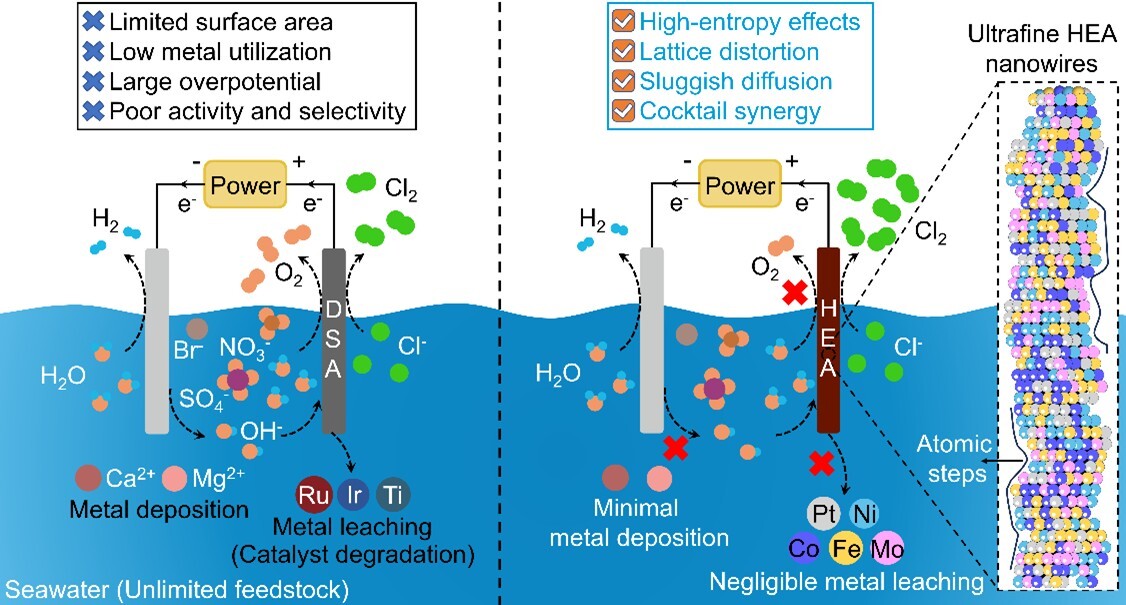
Schematic illustration of direct chlorine electrosynthesis from seawater using 1D PtNiCoFeMo UF-HEANWs for the chlor-alkali industry. (Image by YANG Yongchao et al.)
Contact: ZHAO Shenlong
National Center for Nanoscience and Technology (NCNST)
E-mail: zhaosl@nanoctr.cn




