Recently, a research team led by Prof. ZHAO Shenlong from the National Center for Nanoscience and Technology (NCNST) of the Chinese Academy of Sciences made significant progress in the controllable construction of nanocatalytic sites and their application in coupling anodic aldehyde oxidation with water electrolysis for hydrogen production. The study, titled "Industrial-scale Aldehydes Electrification Via Localized Hydrogen-affinity Engineering", was published in Angewandte Chemie International Edition.
Under the context of the "dual carbon" strategy, zero-carbon industrial parks urgently demand efficient and clean energy conversion technologies. Water electrolysis for hydrogen production serves as a crucial technological path for electricity-hydrogen-chemical coupling. However, its performance heavily relies on the structural design and interface regulation of electrocatalytic materials. To tackle the high energy consumption and low value of the traditional anodic oxygen evolution reaction, the researchers proposed a novel strategy coupling anodic formaldehyde (HCHO) oxidation with water electrolysis, enabling the simultaneous co-production of green hydrogen and high-value chemicals.
Addressing the sluggish dehydrogenation kinetics of traditional copper (Cu)-based catalysts in aldehyde electrooxidation, the researchers developed a computation-guided strategy—localized hydrogen-affinity engineering—to synthesize Rh-decorated Cu hydrogenase single-atom alloy catalyst (Rh1Cu-Hase).
This catalyst achieves a remarkable Faraday efficiency of > 99.3% for formaldehyde conversion at an ultrahigh current density of 500 mA cm−2 with a minimal overpotential of 283 mV, significantly outperforming previously reported electrocatalytic materials. Furthermore, the coupled membrane-free electrolyzer system can operate stably for over 1200 hours at an industrial-level current density of 1000 mA cm−2. This system achieves bipolar hydrogen production and the continuous synthesis of high-purity potassium diformate (KDF). Remarkably, the energy consumption for hydrogen production is reduced to as low as 0.63 kWh Nm−3, demonstrating excellent potential for industrial application.
Through operando characterizations, comparative experiments, and theoretical calculations, the team revealed a Rh-Cu host-guest dual-site synergistic catalytic mechanism, known as the paired dehydrogenation mechanism. The Cu matrix facilitates the adsorption of aldehyde intermediates, while the atomic Rh sites promote electrocatalytic C-H bond cleavage and H-H coupling. This synergy accelerates multi-electron transfer and proton-coupled kinetics, achieving an efficient match between anodic oxidation and cathodic hydrogen evolution. Importantly, this nanocatalytic site construction strategy shows broad universality across various dual-site systems and aldehyde electrooxidations.
This study not only demonstrates a feasible technological route for coupled anodic oxidation and water electrolysis but also provides new insights into the precise design of high-performance nanocatalysts.
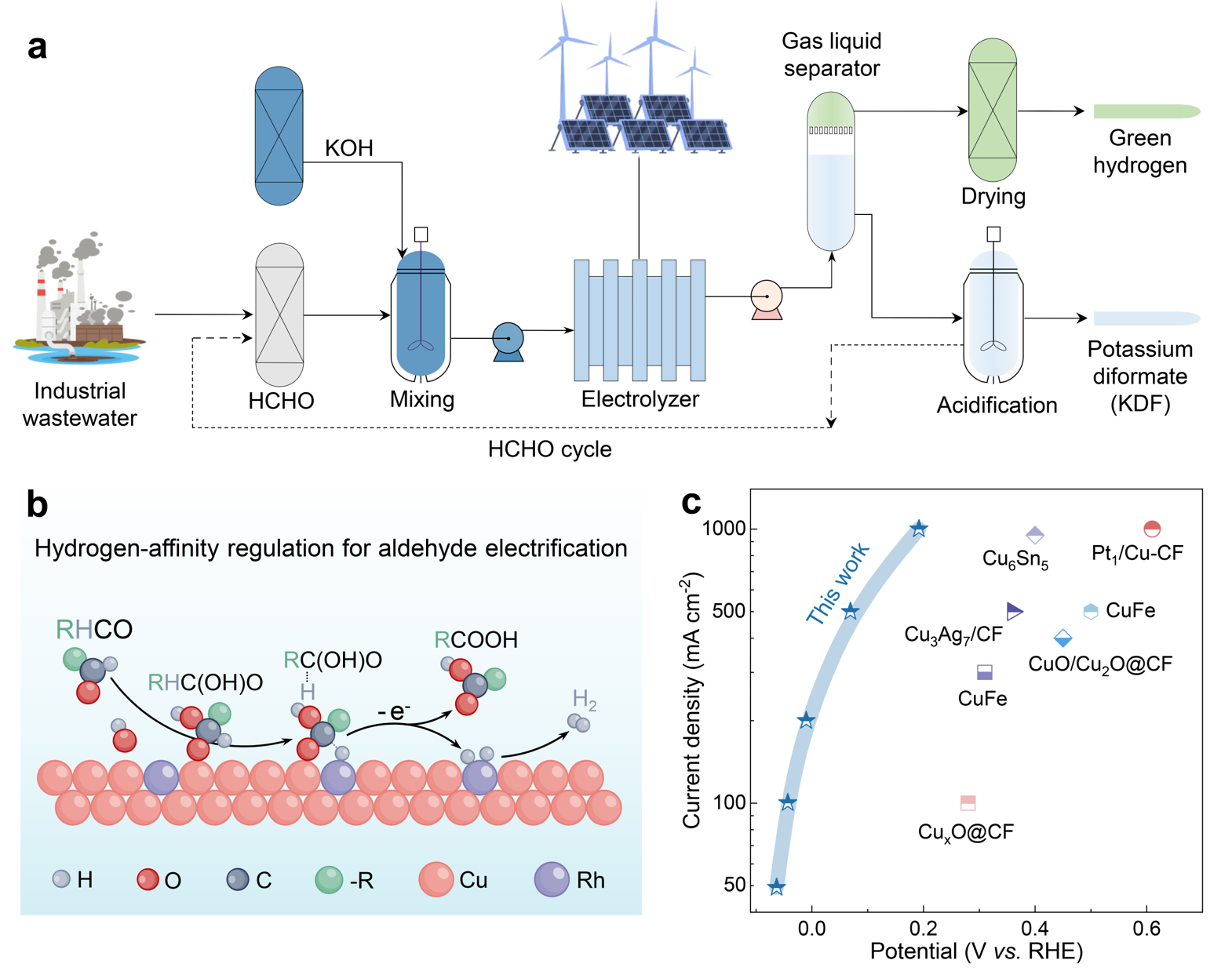
The schematic illustration of the formaldehyde electrification system and corresponding reactions for potassium diformate (KDF) and hydrogen production (Image by SHI Lei et al.)
Contact: ZHAO Shenlong
National Center for Nanoscience and Technology (NCNST)
E-mail: zhaosl@nanoctr.cn




