Pancreatic ductal adenocarcinoma (PDAC), often referred to as the "king of cancers", is notorious for its highly immunosuppressive microenvironment. This "immune desert" state renders conventional immunotherapies largely ineffective. A central challenge in treating PDAC is finding ways to convert these "cold" tumors into "hot" ones, thereby activating the immune system to recognize and attack cancer cells.
In a recent study published in Cell Host & Microbe, a collaborative research team led by Prof. ZHU Motao from the National Center for Nanoscience and Technology (NCNST) and Prof. YANG Yinmo from Peking University First Hospital proposed a solution. By performing 16S rDNA sequencing on tumor tissues from 40 PDAC patients, the team identified Bifidobacterium longum (B. longum) as a dominant, tumor-colonizing probiotic with significant anti-tumor potential.
To transform these "invisible" intratumoral bacteria into recognizable immune targets, the researchers developed an innovative strategy named XenoPro. They utilized low-dose rapamycin to induce "xenophagy"—a selective autophagic mechanism that clears intracellular pathogens. This process forces tumor cells to "digest" the invading B. longum and present its protein fragments on surface MHC-II molecules as novel "bacteria-derived neoantigens".
In orthotopic murine PDAC models, the XenoPro strategy significantly upregulated MHC-II expression on tumor cells, triggering CD4⁺ T cell-dominated immune recognition and profoundly inhibiting tumor growth. Building on this, the team screened 12 highly immunogenic MHC-II-restricted neoantigens from B. longum proteins to construct a therapeutic peptide vaccine (Vax). The combination of XenoPro and Vax demonstrated remarkable synergistic anti-tumor effects, significantly prolonging survival in both orthotopic and liver metastasis PDAC models, as well as showing broad-spectrum potential in colon and melanoma cancer models.
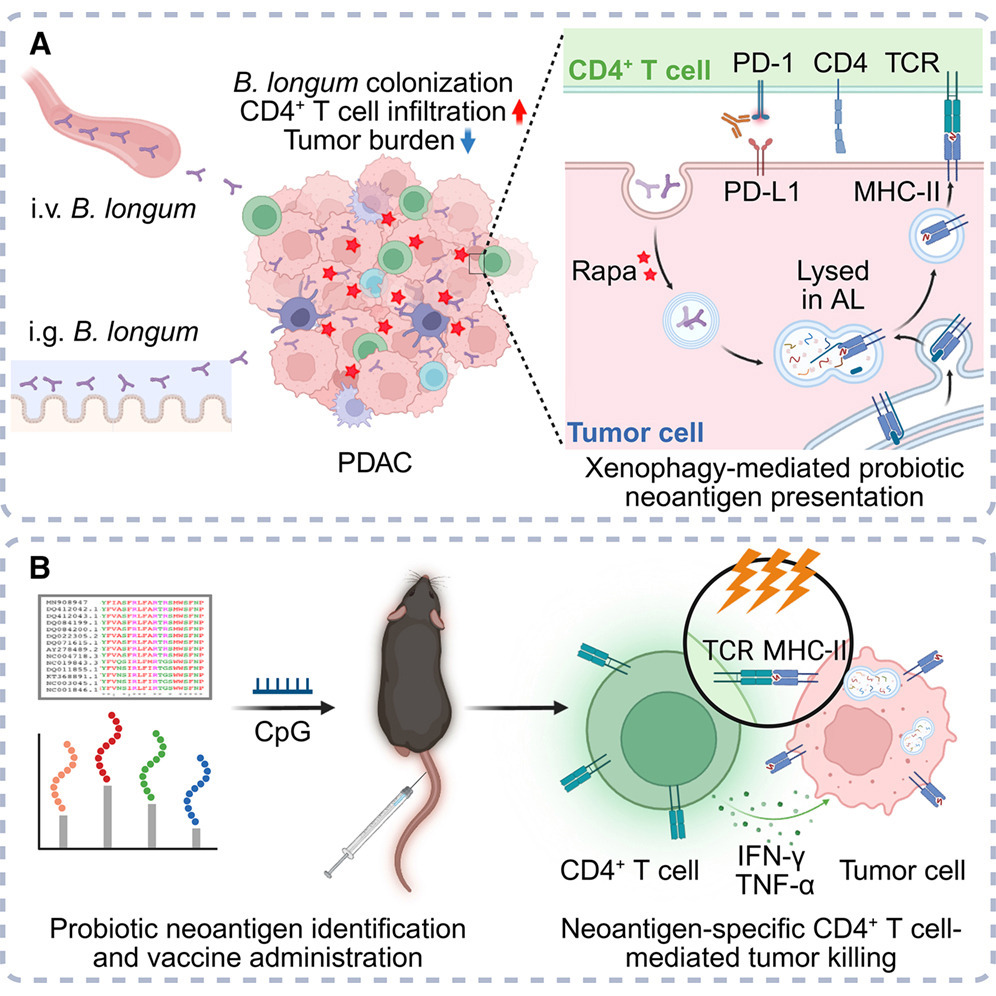
Enhancing its clinical translational value, the study confirmed that oral administration of B. longum achieves comparable anti-tumor efficacy to intravenous injection, with a superior safety profile. Furthermore, combining XenoPro with anti-PD-1 therapy further boosted tumor suppression and optimized the gut microbiome. This xenophagy-based strategy utilizes standardized probiotics as a universal neoantigen pool, successfully bypassing the need for expensive and time-consuming patient-specific neoantigen sequencing, and providing a fresh paradigm for treating immunotherapy-resistant tumors.




