Converting carbon dioxide (CO2) into value-added chemicals like methanol is a critical step toward achieving carbon neutrality and sustainable energy. However, traditional platinum (Pt) nanoparticle catalysts often favor the hydrogenation of CO2 to carbon monoxide (CO), presenting a significant challenge for synthesizing value-added methanol efficiently.
In a recent study published in the Journal of the American Chemical Society, a research team led by Prof. LI Guodong from the National Center for Nanoscience and Technology (NCNST) developed a novel catalytic system to overcome this long-standing limitation. The researchers successfully reported a sulfonic acid group docking strategy to achieve the gram-scale synthesis of monodispersed Pt clusters. These clusters are precisely confined within the cavities of a double sulfonated zirconium terephthalate MOF, known as UiO-66.
To modify the catalytic behavior and prevent the typical CO byproduct formation, the team utilized strategically positioned sulfonic acid groups within the MOF cavities as precise anchors for the Pt clusters, which is also applicable to other precise metal cluster.
This tailored nano-architecture drastically alters the reaction pathway. The resulting catalyst demonstrates exceptional performance, enabling the efficient hydrogenation of CO2 directly to methanol. Remarkably, it achieves nearly 100% methanol selectivity at a low temperature of 60 °C and sustains highly stable catalytic performance.
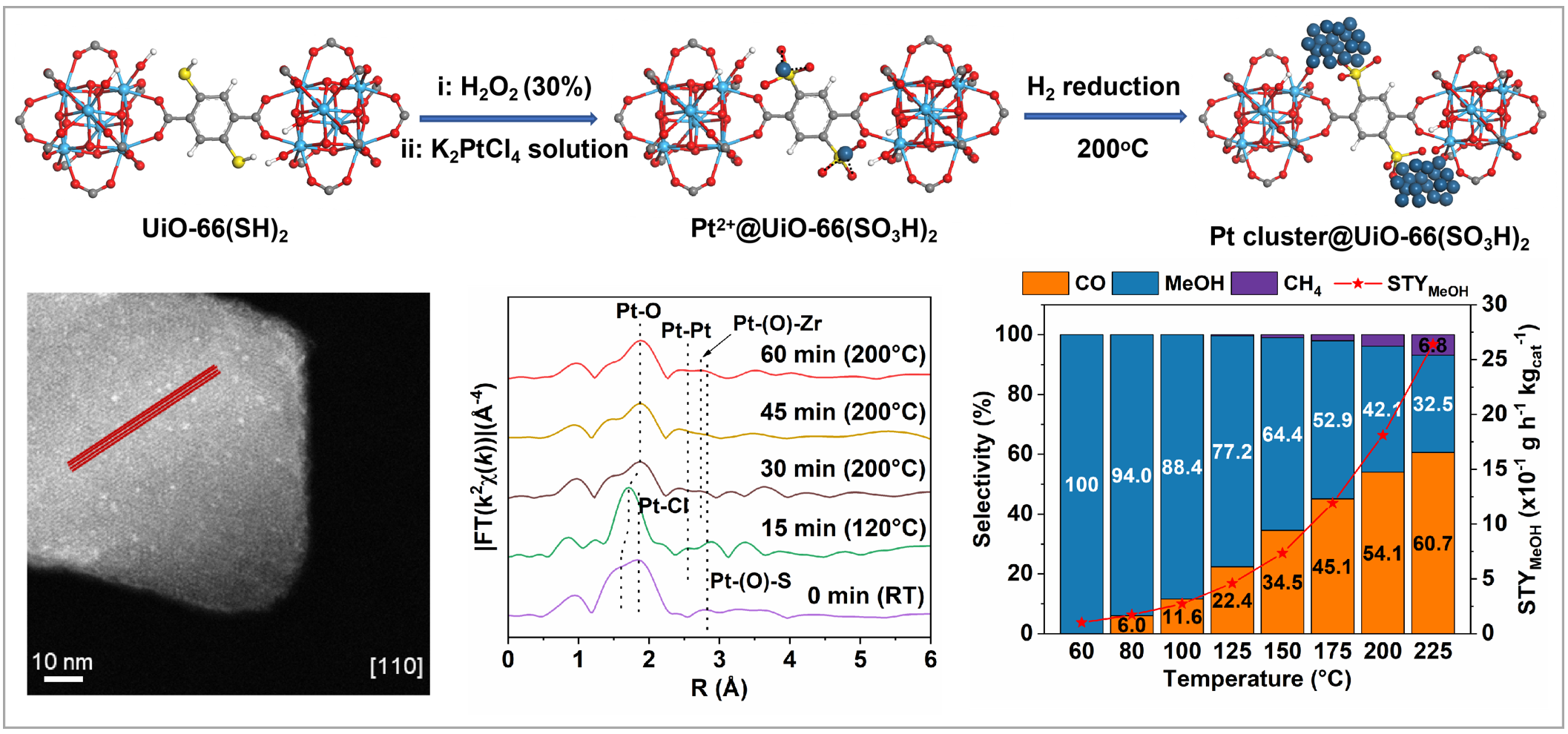
This study not only resolves the selectivity issue of Pt-based catalysts in CO2 hydrogenation but also provides a rational design paradigm for confining metal clusters in porous materials for advanced energy applications.




