Advances and challenges of CAR-T immunotherapy
CAR-T immunotherapy has revolutionized the treatment of hematological malignancies, particularly B-cell lymphoma. However, while CAR-T cells effectively kill tumor, they can also trigger the release of large quantities of inflammatory cytokines from immune cells, particularly macrophages. These excessive cytokines can lead to severe and life-threatening symptoms, including high fever, hypotension, weight loss, vascular leakage, organ failure and even death. These symptoms induced by CAR-T cells are known as cytokine release syndrome (CRS) or inflammation storm.
CRS characteristics
CRS is frequent occurrence with CAR-T cell therapy, yet its precise diagnosis, timing, and severity assessment have proven challenging due to the absence of specific physiological index. Current intervention measures were generally given after CRS occurrence. Although the timely treatment can alleviate CAR-T induced CRS, some symptoms that have already occurred like the vascular leakage can cause irreversible damage to patients.
Drug administration issues
Although the FDA has approved the interleukin-6 (IL-6) blockade monoclonal antibody for the management of CAR-T induced CRS, these blockade antibodies still face some issues, including systemic toxicity and short duration of effect. Furthermore, IL-6 molecules themselves play important physiological roles under normal conditions, such as promoting B-cell proliferation. Therefore, injecting blockade antibodies in advance or when CRS doesn’t occur can affect the normal levels of IL-6 and related functions.
The “Interleukin-6 Sponge (IL6S)” strategy
Based on CRS characteristics and IL-6 antibody issues, the research team, led by LIANG Xingjie and WU Yan at National Center for Nanoscience and Technology, proposed the “IL-6 sponge (IL6S)” strategy for the prevention of CAR-T cell-induced cytokine release syndrome (CRS). In this study, IL-6 antibody was chemically linked to a temperature-sensitive hydrogel. The obtained IL-6 antibody-hydrogel conjugate (IL-6 sponge) is subcutaneously injected before CAR-T cells infusion. When CAR-T cells induce an inflammatory cytokine release storm, the injected IL-6 sponge can adsorb IL-6 in real-time and controllable manner, thereby preventing CRS-related symptoms.
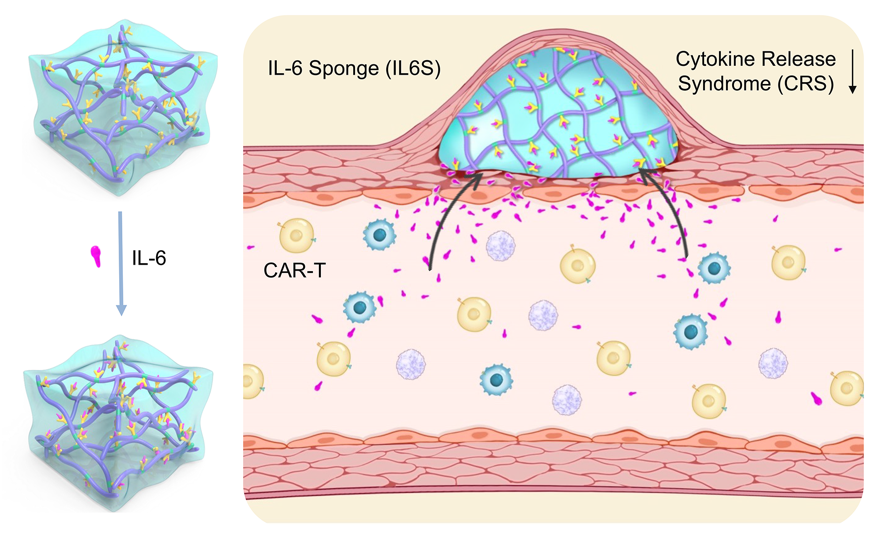
Figure 1. Schematic diagram of IL-6 sponge in prevention of CAR-T induced cytokine release syndrome. (Image by LIANG Xingjie et al)
IL-6 sponge adsorbs IL-6 in a responsive manner
By adjusting the antibody-to-hydrogel ratio, the subcutaneously injected IL-6 sponge does not affect the normal levels of IL-6. It only adsorbs IL-6 when its concentration begins to rise or exceeds normal levels. This responsive adsorption can not only simplify disease assessment but also convert CRS management from traditional blockade alleviation to effective prevention.
IL-6 suppressed CAR-T induced CRS without compromising antitumor effect of CAR-T cells
The researchers validated the therapeutic effects of the IL-6 sponge on various CAR-T induced CRS models. The IL-6 sponge not only effectively suppressed the elevation of various inflammatory cytokines but also greatly alleviated CRS-related symptoms, and improved the survival rate of animal model. Moreover, the IL-6 sponge did not affect the antitumor efficacy of CAR-T cells in vivo, demonstrating its excellent safety performance.
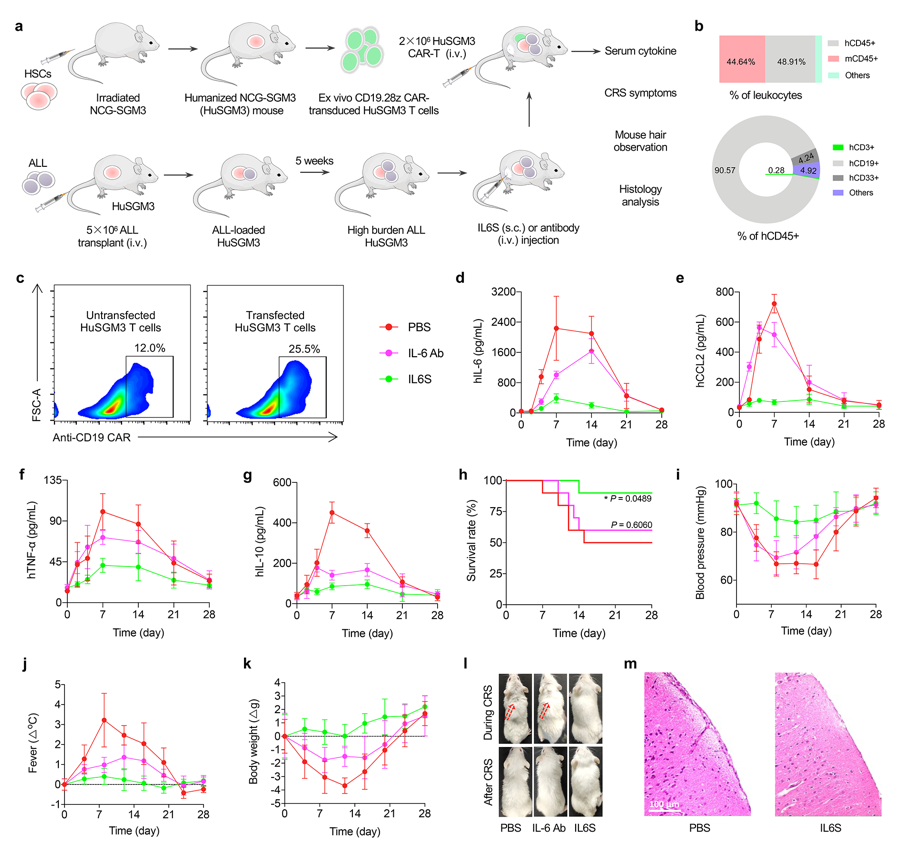
Figure 2. IL-6 sponge suppressed CAR-T induced CRS in a humanized NCG-SGM3 mouse model. (Image by LIANG Xingjie et al)
Contact:
LIANG Xingjie
National Center for Nanoscience and Technology
E-mail:liangxj@nanoctr.cn




